Medication Absorption Risk Checker
Risk Level
Detailed analysis will appear here.
Disclaimer: This tool is for educational purposes only and does not replace professional medical advice.
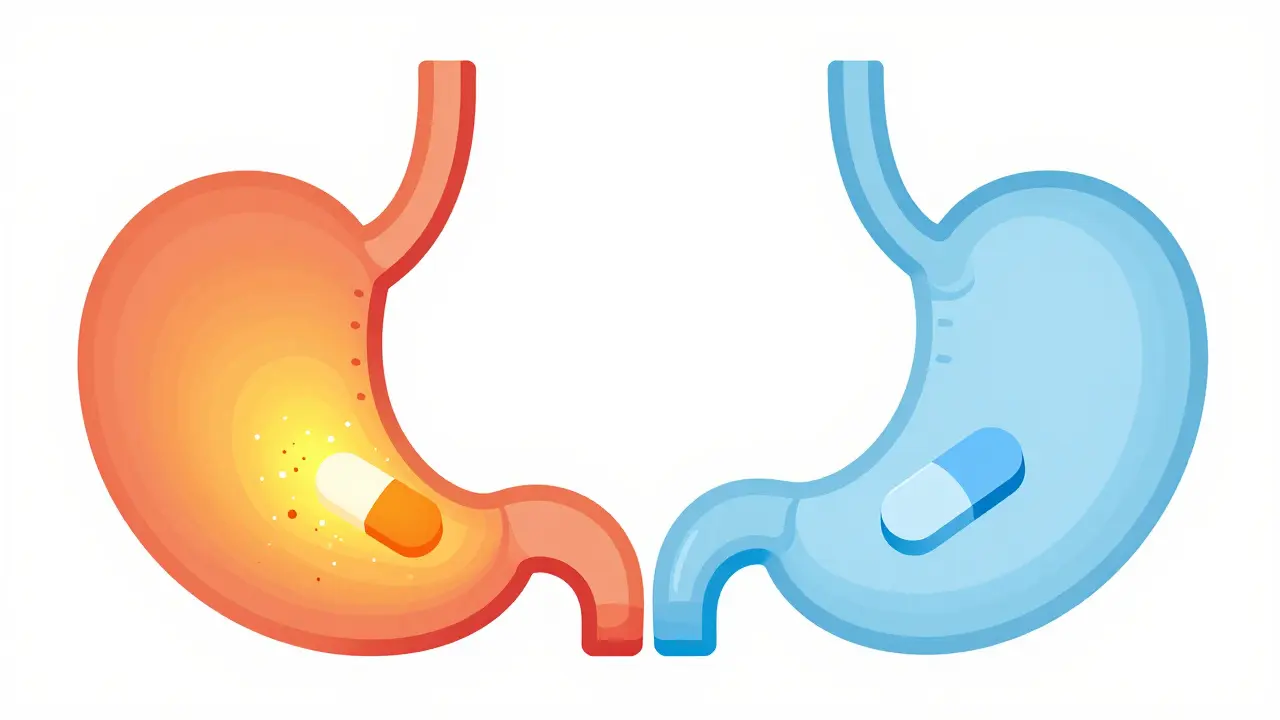
Imagine taking a life-saving medication every day, only to find out months later that it wasn't working because of a simple over-the-counter pill you took for heartburn. It sounds like a fluke, but for millions of people, it's a real clinical risk. Many of us reach for a tablet to stop acid reflux without realizing that changing the chemistry of our stomach can essentially "lock" other medications out of our bloodstream. When you raise the pH of your stomach, you aren't just stopping the burn; you're potentially changing how your body absorbs everything from HIV treatments to antifungal meds.
| Key Point | What You Need to Know |
|---|---|
| The Core Issue | Raising stomach pH can make certain drugs less soluble, preventing them from entering the bloodstream. |
| Highest Risk | "Weak base" drugs (like Atazanavir or Ketoconazole) are the most vulnerable to absorption failure. |
| PPI vs H2RA | PPIs (like Omeprazole) have a stronger, longer-lasting effect on pH than H2 blockers (like Famotidine). |
| The Solution | Staggered dosing or switching to pH-independent formulations often solves the problem. |
The Chemistry of the Stomach and Drug Solubility
To understand why this happens, we have to look at the stomach's natural state. Normally, your stomach is an acidic powerhouse with a pH ranging from 1.0 to 3.5. This environment isn't just for digesting food; it's critical for breaking down pills. Acid-reducing medications is a class of drugs, including Proton Pump Inhibitors (PPIs) and H2-receptor antagonists (H2RAs), designed to decrease gastric acid production. When these drugs do their job, they push that pH up to 4.0 or even 6.0.
Here is where the physics comes in. Most drugs are either "weak bases" or "weak acids." About 70% of oral meds are weak bases. In a highly acidic stomach, these bases become ionized, which makes them dissolve easily. But when a PPI raises the pH, these drugs stay in a non-ionized form. If a drug can't dissolve, it can't be absorbed. While most of the actual absorption happens in the small intestine, the process usually starts with dissolution in the stomach. If the drug never dissolves, it simply passes through your system without ever helping you.
Comparing PPIs and H2 Blockers: Which Is Riskier?
Not all acid reducers are created equal. You'll generally find two main types: Proton Pump Inhibitors (PPIs) and H2-receptor antagonists (H2RAs).
PPIs, such as Omeprazole , are the heavy hitters. They irreversibly shut down the acid pumps in your stomach lining, keeping the pH high for 14 to 18 hours a day. Because the acid suppression is so profound and sustained, the risk of drug interaction is much higher. In contrast, H2 blockers (like Cimetidine) block histamine receptors and typically keep the pH elevated for only 8 to 12 hours. Research shows that PPIs can reduce the absorption of pH-dependent drugs by 40-80%, whereas H2RAs usually only cause a 20-40% drop.

High-Risk Medications: The Danger Zone
Some drugs are far more sensitive to pH changes than others. If you are taking a medication with a "narrow therapeutic index"-meaning a small difference between a dose that works and a dose that fails-even a slight dip in absorption can be catastrophic. Atazanavir is an HIV protease inhibitor that serves as the most extreme example. When taken with a PPI like Lansoprazole, its absorption can plummet by up to 95%. This can lead to viral rebound, where a patient's viral load spikes because the medicine simply isn't getting into their system.
Other high-risk examples include:
- Dasatinib: Used for leukemia; absorption can drop by 60%, often requiring a dose increase.
- Ketoconazole: An antifungal that can become almost entirely ineffective if the stomach isn't acidic enough.
- Enteric-coated tablets: These are designed to dissolve only in the alkaline environment of the intestine. If your stomach pH is too high, these coatings may dissolve too early, potentially irritating your stomach or destroying the drug before it reaches the right spot.
Real-World Consequences of Poor Absorption
This isn't just theoretical chemistry; it has real impacts on patients. There are documented cases in clinical databases where people experienced complete treatment failure. One retrospective study of over 12,000 patients found that those using PPIs while taking Dasatinib had 37% higher treatment failure rates. It's a hidden danger because the patient feels like they are following their prescription perfectly, but the acid-reducing medications are acting as a barrier.
Even blood pressure medications can be affected. Some users have reported their readings climbing by 20 points after starting a PPI, simply because their body wasn't absorbing their hypertensive medication efficiently. This is why the FDA has recently pushed for updated labeling on dozens of drugs to warn against concomitant use with acid reducers.

How to Manage Interactions and Stay Safe
If you need both an acid reducer and a pH-dependent medication, you don't always have to choose one or the other. There are practical ways to mitigate the risk. The most common strategy is staggered dosing. For many weak base drugs, taking the medication 2 hours before the acid reducer can help. However, this isn't a perfect fix; it only reduces the interaction magnitude by about 30-40% in some cases.
A more effective approach is often a total switch in medication. Your doctor might suggest:
- Switching to a pH-independent formulation: Newer drugs are being developed specifically to bypass the need for stomach acid for dissolution.
- Using antacids: These have a much shorter duration of action than PPIs, making it easier to time your other meds around them.
- Deprescribing: Many people take PPIs long-term without a clear medical need. Reducing or stopping these meds can restore your natural gastric pH and fix absorption issues.
The best line of defense is a thorough medication review with a pharmacist. Pharmacist-led reviews have been shown to reduce inappropriate acid-reducer co-prescribing by over 60%. If you're on a complex regimen, ask your provider if your medications are "pH-dependent."
Do all acid-reducing medications cause these problems?
No, but most do to some extent. PPIs are the most problematic because they provide the strongest and longest acid suppression. H2 blockers have a milder effect, and simple antacids have the shortest impact. However, any drug that significantly raises your stomach pH can potentially interfere with the absorption of weak base medications.
Can taking an acid reducer actually help some drugs absorb better?
Yes, in rare cases. Weakly acidic drugs (like aspirin) can actually see a modest increase in solubility when the pH is higher. For example, some medications like dasiglucagon might see a 15-20% increase in absorption, though this rarely requires a change in dosage because the effect is usually minimal.
What is the "2-hour rule" for dosing?
The 2-hour rule suggests taking your pH-dependent medication at least two hours before your acid reducer. This allows the drug to dissolve in the naturally acidic environment of the stomach before the medication kicks in to raise the pH. While helpful, it may not be sufficient for high-risk drugs like Atazanavir.
Why does the small intestine not just handle the absorption?
While it's true that about 90% of absorption happens in the small intestine due to its massive surface area, most pills must first dissolve (disintegrate) in the stomach. If a pill stays as a solid clump because the pH is too high, it cannot be absorbed efficiently by the time it reaches the small intestine.
Should I stop taking my PPI if I'm worried about interactions?
Never stop a prescribed medication without talking to your doctor first. Acid reducers are vital for treating GERD and peptic ulcers. Instead, ask your doctor if there is an alternative acid reducer or if you can adjust the timing of your doses to avoid interactions.
Next Steps for Patients and Caregivers
If you are currently taking a PPI or H2 blocker along with other chronic medications, here is a simple game plan:
- Audit your meds: Make a list of everything you take, including over-the-counter supplements.
- Check for "Weak Bases": Ask your pharmacist if any of your medications are known to be pH-dependent.
- Evaluate the need: If you've been on a PPI for years, ask your doctor if a "deprescribing" plan is appropriate for you.
- Monitor efficacy: If you start an acid reducer and notice your other medications aren't working as well (e.g., blood pressure rises, or symptoms return), report it to your doctor immediately.

Victor Parker
April 10, 2026 AT 13:43Big Pharma just wants us on these pills forever so they can keep us sick and dependent 🙄
The government knows exactly what's happening and they just let it slide so we keep buying the "solutions" to the problems they created in the first place! 😱